This web page was produced as an assignment for Bioinformatics 490-006, an undergraduate and graduate student course at Southern Illinois University Edwardsville.


DMD
Dystrophin protein encoding gene
WHAT IS DYSTROPHIN
What Does It Do?
The Full Story (And a Large One At That)
The dystrophin gene ecodes for a large protein that is a part of the dystrophin-glycoprotein complex (DGC). This complex bridges the inner cytoskeleton and extracellular matrix, and is responsible for mechanical stabilizing and signaling in muscle tissue. Deletions, duplications, and point mutations in this gene may result in Duchenne and Becker Muscular Dystrophy, and Cardiomyopathy
Located on chromosome xp21.1-p21.1 the dystrophin gene spans over 2.3 million base pairs and is the largest gene in the human genome. This gene is home to 2,220,242 nucleotides. 79 exons, and 32 different transcript isoforms. Most of these variants have 40 amino acids.
Fig. 1 ideograms shows the chromosome DMD is located in on the X chromosome. Clicking on the picture will take you to NCBI's Genome Data Viewer.

Many Forms
DMD comes in many isoforms and many deleterious mutation types have been documented
The picture on the left shows a circular neighbor joining tree of the 30 reference protein sequences of the DMD gene sourced from NCBI.
The picture to the right shows the 30 mRNA reference sequences, many of which have mutations causing Becker Type Muscular Dystrophy

Transcript Expression
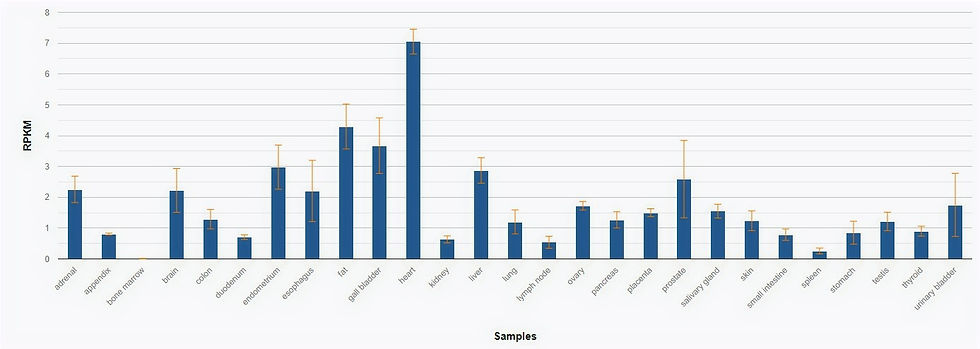
In 2008, RNA-seq was performed on 95 individuals to test 27 samples and identify any tissue specificity. Results were measured in Reads Per Kilobase of transcript, per Million mapped reads. Fig. 2 shows that the DMD gene is largely expressed in the heart tissue.
Fig 2. RNA-seq table of DMD expression in various tissue samples measure in RPKM.
Fagerberg, L., Hallström, B. M., Oksvold, P., Kampf, C., Djureinovic, D., Odeberg, J., … Uhlén, M. (2013). Analysis of the Human Tissue-specific Expression by Genome-wide Integration of Transcriptomics and Antibody-based Proteomics. Molecular & Cellular Proteomics, 13(2), 397–406. doi:10.1074/mcp.m113.035600
Fig. 3 Protein-protein interactions taken from BioGrid.
Protein-Protein interaction for Dystrophin
fig. 3 is a visual representation of the 64 protein-protein interactions with dystrophin. There is 64 known interactors with 87 different kinds of interactions.
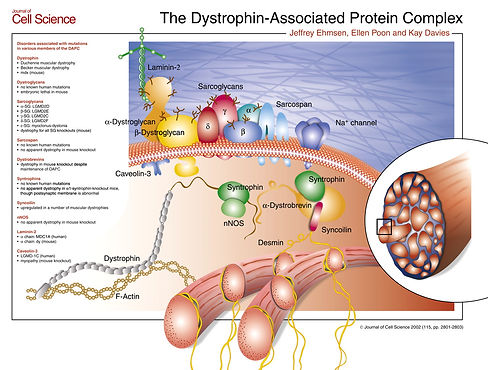
Many of these proteins come together to form the dystrophin-associated glycoprotein. This is shown in fig. 4
Journel of Cell Science 2002 (115, pg, 2801-2803)
Fig. 5 shows that the DMD gene is entirely intra cellular, and only a part of the larger comlex linking the cytoskeleton to the extra cellular matrix
fig. 4 The Dystrophin-Associated Protein visual trom the Journel of Cell Science displaying how each protein in the complex interacts.

Fig. 5 a protein map of DMD show it has no intramembrane domains, and is located only inside the cellular matrix.
REVIEW OF THE PAPER "DUCHENNE MUSCULAR DYSTROPHY GENE EXPRESSION IN NORMAL AND DISEASED HUMAN MUSCLE"
Published in the article Science in 1988
Authors: M. ORONZI Scorr, J. E. SYLvEsTER, T. HEimAN-PATrERSON, Y.-J. SHI, W. FIELES, H. STEDMAN, A. BURGHES, P. RAY, R. WORTON, K. H. FISCHBECK
In Situ hybridization was performed on human muscle of a healthy individual, and of individuals displaying muscular dystrophy. Researches found a lower expression of dytrophin in muscles of patients with a mutated DMD gene. However, there was still faint expression, showing that none of the patients used in this study had a complete null mutation, and that there was some increased expression compensation.